|
 |
Release Date: June 15, 2012 |
EDUCATIONAL OVERVIEW Addressing Barriers to Adult Immunization: To improve overall adult immunization rates, it is critical to expand PCP knowledge and competence regarding vaccine-preventable diseases and recommended vaccines. Building healthcare provider awareness of current immunization guidelines as well as the benefits of immunization is critical to improving vaccination rates. PCPs can lead the way in these efforts by educating their patients about vaccine-preventable diseases and taking proactive measures within their clinical practice to ensure patients are vaccinated when indicated. TARGET AUDIENCE LEARNING OBJECTIVES
EDUCATIONAL FORMAT Through evidence-based presentations and faculty discussions, this 10-module online activity, provides evidence supporting the importance of adult vaccination and describes the current recommendations. It also addresses the challenges PCPs may face in promoting adult vaccination and identifies strategies that can be implemented in clinical practice to assist their immunization efforts. This online activity has 10 modules. MODULE 1: Pneumococcal Disease MODULE 2: Influenza MODULE 3: Pertussis MODULE 4: Herpes Zoster MODULE 5: Don’t Neglect the Other Adult Vaccines MODULE 6: Adult Immunization: Role of the Clinician MODULE 7: The Medical Home: Utilizing a Multidisciplinary Approach to Adult Immunization MODULE 8: Quality Improvement and Meeting the Managed Care Requirements in Adult Immunization MODULE 9: Utilizing a Combination of Interventions in Improving Adult Immunization MODULE 10: Role of the Pharmacist in Adult Immunization Note: 1. This enduring material is based on a series of live workshops presented between December 2011-April 2012. 2. If you have received credit for attending the live workshop by the same name, you are not eligible to receive credit for this enduring material. 3. Note that The Task Force on Community Preventive Services recommendations presented in Module 9 have been updated since this activity was released. Current recommendations on combination and standalone interventions are available at:
|
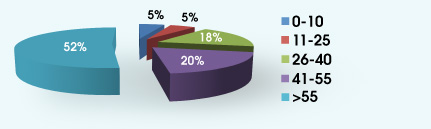
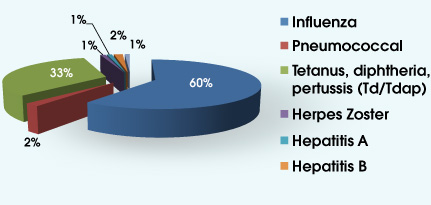
AUDIENCE RESPONSE RESULTS 1. How many patients do you see each week?
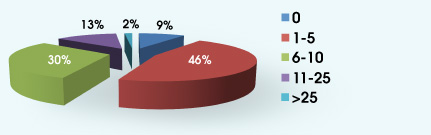
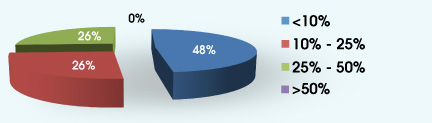
4. Approximately what percentage of your patients
|
CE Accreditation NFID designates this enduring material for a maximum of 2.0 AMA PRA Category 1 Credits™. Physicians should claim only the credit commensurate with the extent of their participation in the activity. Method of Participation and Instruction for Credit
For questions, please contact cme@nfid.org. Disclosures Faculty Debra A. Goff, PharmD serves as a consultant/advisor to Cubist, Forest, Merck & Co., Inc., and Optimer. She serves as speaker for Merck & Co., Inc. Michael D. Hogue, PharmD serves as a consultant/advisor for Pfizer Inc. and as a speaker for Merck & Co., Inc. Robert H. Hopkins, Jr., MD has no relevant financial relationships to disclose. Patrick Joseph, MD has no relevant financial relationships to disclose. Donald E. Low, MD has no relevant financial relationships to disclose. George G. Zhanel, PharmD, PhD has received grants for clinical research from Abbott, Achaogen, Affinium, Astellas, AstraZeneca, Cangene, Cubist, Forest/Cerexa, Merck & Co., Inc., and Pfizer Inc.
|
Planning Committee Members Marla Dalton (NFID) has no relevant financial relationships to disclose. Susan J. Rehm, MD (NFID) serves as an advisor or consultant for Merck & Co., Inc. and Pfizer Inc. and as a speaker for Genentech. All other employees of NFID and Vemco MedEd have no relevant financial relationships to disclose. Disclaimer Hardware/Software Requirements Software/Hardware Connection Speed System Check Copyright Statement Privacy Policy Sponsorship
|
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
  |